chemical Dynamics
Little would you expect that chemical kinetics would at all relate to taste, but to your surprise, the reactions taking place between different food molecules themselves, and water/ saliva, can be classified as zero, first and higher order reactions.
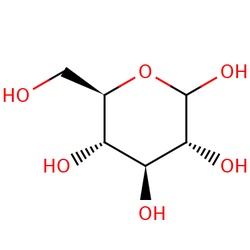 D-glucopyranose
D-glucopyranose
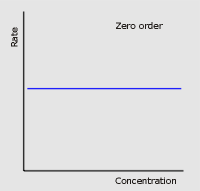
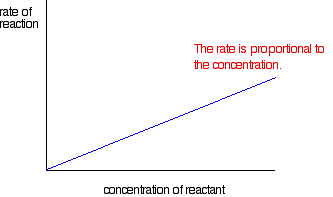
For example, when an enzyme (biological catalyst) found in the body called glucoamylase, begins its work in the mouth to reduce excess amounts of starch by turning it into simpler sugars such as glucose for more useful energy, a zero order reaction takes place. This means that the rate of the reaction is completely independent of the change in initial concentration of the starch as a reactant. So if the concentration doubled or tripled, the rate would remain the same. That is why some larger carbohydrates do not taste sweet right away, since they have not yet been reacted and broken down into their simpler sugars. A visual representation of this relationship can be seen in the graph on the left. First order reactions on the other hand mean that the initial concentration is multiplied to the exponent of one. Therefore, if the initial concentration of one of the reactants were to double, so would the rate, and if it were to triple, the rate would too. This is exemplified when D-glucopyranose (a cyclic sugar) does a change in rotation or formation, while dissolving in saliva. As the concentration of the sugar in the water/saliva increases, the rate of the reaction does the same proportionately. A visual representation of this relationship can be seen in the graph on the right.
|
When calculating the rates of such reactions that the chemicals undergo when reacting with and before latching with receptors, the rate law equation can be used. This equation is defined as “the relationship among rate, the rate constant, the initial concentrations of reactants, and the orders of reaction with respect to the reactants.”
|
It is commonly expressed as: r = k [X]^m [Y]^n
r symbolizing the rate of reaction, k symbolizing the reaction constant, X and Y symbolizing the initial concentrations of each reactant and, m and n symbolizing their orders of reaction. |
what affects these rates of reaction?
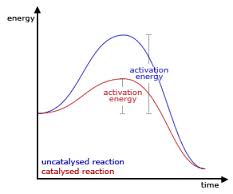
There are five factors that can possibly affect the rate of a reaction for molecules being interpreted as taste. First of all, the chemical nature of the reactants play a huge role in the rate. This is exemplified as simpler compounds such as sodium chloride react much faster than molecules of a greater complexity including glucose. This is simply due to the fact that the more complex the molecule, the greater the number of bonds that must be broken in order to produce the product. Secondly, as stated before, depending on the order of the reaction the concentration has a huge impact on the speed at which it takes place. Thirdly, as can be predicted, the temperature makes a considerable difference in means of the speed of a reaction. The higher the temperature, the faster a reaction will proceed because there is more energy to help break the bonds. Fourthly, the presence of a catalyst can be used to either speed up or slow down the rate of reaction. For example, soda crackers are made out of starch that does not taste sweet until after it has been chewed for a while. At this point, amylase, which is a catalyst found in saliva, has broken the starch into smaller sugar molecules and sped up the reaction. Last but not least, the surface area of the reactants affects the rates in which they are reacted. The greater the surface area, the faster the reaction. For example, powdered sugar dissolves quicker in your mouth than does cubes of sugar because more of it can react at once to stimulate the taste cells.