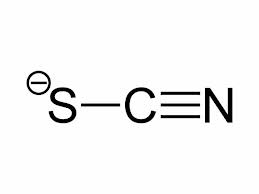Saliva
Saliva, as mentioned earlier is one of the key components of the gustatory system because it reacts with food molecules to dissolve and break them up into their simplest chemical structures in order to be considered a tastant and react with a receptor. In general, there are eleven major components that make up saliva, all with different chemical features and purposes. Some of which primarily include, electrolytes, mucus, antibacterial compounds, enzymes, cells, and its main component being water. With the combination of all of these components, saliva can fulfill its purpose of lubrication, digestion, protection and most importantly, taste. Here is a general break down of each component:
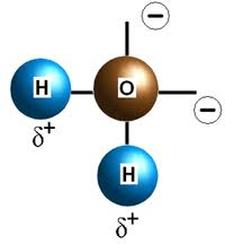
Water is a very unique molecule. As the oxygen atom must gain two electrons to obtain a full octet, and each of the two hydrogen atoms must lose one election to similarly obtain a complete outer shell, two polar covalent bonds are formed between the oxygen atom and the two hydrogen atoms. Due to the fact that oxygen has a higher electronegativity than that of hydrogen, the oxygen atom carries a partial negative charge, while the hydrogen atoms carry partial positive charges because the elections being shared in between the atoms spend more time around the oxygen atom. Furthermore, its geometrical shape is considered to be V-shaped, with a tetrahedral electron pair arrangement, which also contributes to its polar nature. Since water is a very polar molecule and has a great ability to dissolve and dissociate a variety of different compounds, it is known as the universal solvent and consequently compensates for majority of the make up of saliva which aids with the gustatory system.
On the other hand, water can be interpreted as the key to the lock, in terms of the tasting being able to access the receptor because the interactions are transmitted through water.
On the other hand, water can be interpreted as the key to the lock, in terms of the tasting being able to access the receptor because the interactions are transmitted through water.

Electrolytes are practically charged ions that are found in body fluids to maintain voltages across cells which are needed to conduct messages and support chemical reactions. The electrolytes in the saliva and their various concentrations include:
"- positive sodium ions, 2-21 mmol/L
- positive potassium ions, 10-36 mmol/L
- positive calcium ions, 1.2-2.8 mmol/L
- positive magnesium ions, 0.08-0.5 mmol/L
- negative chloride ions, 5-40 mmol/L
- negative bicarbonate ions, 25 mmol/L
- negative phosphate ions, 1.4-39 mmol/L" ("what chemical is saliva made up of?.")
"- positive sodium ions, 2-21 mmol/L
- positive potassium ions, 10-36 mmol/L
- positive calcium ions, 1.2-2.8 mmol/L
- positive magnesium ions, 0.08-0.5 mmol/L
- negative chloride ions, 5-40 mmol/L
- negative bicarbonate ions, 25 mmol/L
- negative phosphate ions, 1.4-39 mmol/L" ("what chemical is saliva made up of?.")
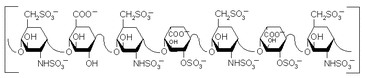
Furthermore, a certain percentage of saliva is made up of mucus, a slimy material that is secreted in fluids and coats many epithelial surfaces. The role of the mucus does not directly contribute to the sensation of taste however, it has an important purpose in binding food together into a bolus so that it can be digested. Mucus is primarily composed of mucopolysaccharides and glycoproteins.
Mucopolysaccharides and glycoproteins, such as heparin (see picture to the left), are chains composed of D-glucosamine and uronic acid molecules linked together.
Mucopolysaccharides and glycoproteins, such as heparin (see picture to the left), are chains composed of D-glucosamine and uronic acid molecules linked together.
Following, there are three primary antibacterial compounds that are found to be a part of saliva.
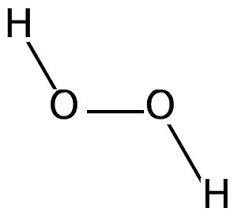
The first of the three being thiocyanate (SCN−), a negative ion/ salt of thiocyanate acid. The second is hydrogen peroxide (H2O2), a polar molecule, and the third is secretory immunoglobulin A, which is an antibody produced through the mucus linings to fight bacteria. See the thiocyanate ion on the left and the polar hydrogen peroxide molecule on the right.
The first of the three being thiocyanate (SCN−), a negative ion/ salt of thiocyanate acid. The second is hydrogen peroxide (H2O2), a polar molecule, and the third is secretory immunoglobulin A, which is an antibody produced through the mucus linings to fight bacteria. See the thiocyanate ion on the left and the polar hydrogen peroxide molecule on the right.
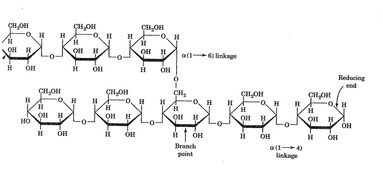 the molecular structure of amylase consists of many hydroxyl groups, thus giving it
properties such as a high melting point and solubility in water do to its polarity.
the molecular structure of amylase consists of many hydroxyl groups, thus giving it
properties such as a high melting point and solubility in water do to its polarity.
Next, our saliva is composed of various types of enzymes that are biological catalysts, responsible for speeding up and initiating many of the reactions that occur between the food molecules and the receptors before the taste can be interpreted.
"-α-amylase which is in charge of intitating the break down of starches and fats into simpler sugars (thus resulting in the sweet flavour)
-Lingual lipase which does not become activated until it enters into the stomach
-Antimicrobial enzymes which is in charge primarily of killing bacteria
-Lysozyme that must break down bacteria before entering into the digestive system
-Salivary lactoperoxidase which catalyses the production of an antibacterial compound from the reaction between hydrogen peroxide and thiocyanate
-Lactoferrin which acts as a guard for harmful microbes and also helps to maintain our immune system
-Immunoglobulin A which is a antibody
-Proline-rich proteins that have multiple functions including the maintenance of the enamel on our teeth and lubrication" ("what chemical is saliva made up of?.")
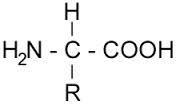 Amino group on the left and carboxylic group on the right, therefore making the molecules extremely polar and giving it a high melting point as a result of the numerous bonds that must be broken
Amino group on the left and carboxylic group on the right, therefore making the molecules extremely polar and giving it a high melting point as a result of the numerous bonds that must be broken
Last but not least, there has been approximately 500 million bacterial cells found per mL of saliva. Majority of these tend to be classified as either amines or organic acids which in turn give the saliva a slight foul smell and taste. Recall that amines are organic molecules with an amino group attached to the end, whereas carboxylic acids are organic molecules with a carboxyl group on the end.