|
|
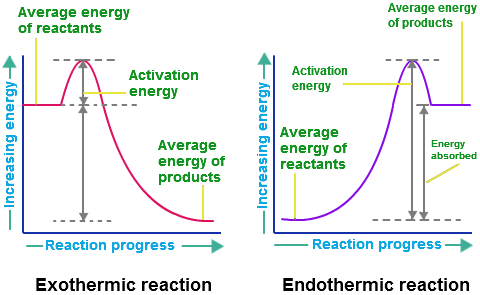
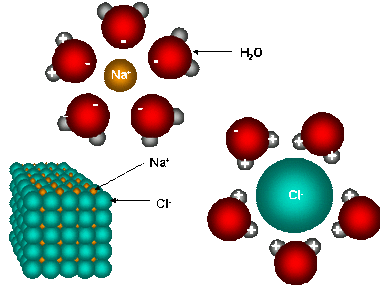
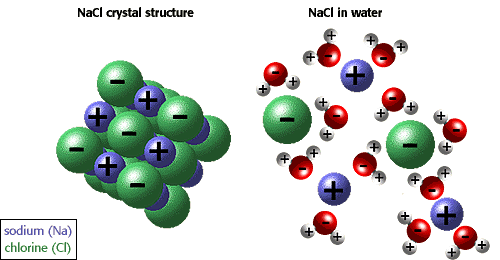
Regardless of the reaction, some sort of energy source must be provided so that the recognition between the tastant and the receptor can occur. For majority of the reactions that take place between molecules before being interpreted, the source of energy that is responsible for disrupting the bonds that hold it together, comes from the structure of the water molecule which is found in saliva. As a result, when substances are dissolved in water, a process known as dissolution occurs as energy is released while the bonds are broken apart into their ions. Due to the fact that water is highly polar and has a greater force holding together its lattice structure, it is the water molecule that breaks the bonds of the other molecule (such as sodium chloride), and because energy is released during the process, the reaction is considered to be exothermic. In order for this reaction to even commence, a certain amount of energy known as the “activation energy” must be obtained to break the bonds, before an even greater amount of energy can be released. Furthermore, because these reactions are exothermic, the enthalpy change of the system is negative.
For example, if the dissolution of sodium chloride in the saliva to stimulate a salty taste were to be contained within a small bubble, defined as the system and the rest of the mouth were considered to be the surroundings, the change in energy of the system which is known as the enthalpy change would decrease and thus be represented by a negative sign. Consequently, the energy in the system would decrease and the energy in the surroundings would increase because the reaction would be giving off energy. However, the enthalpy change is accounted for in relation to the system so it would be described as a loss of energy, rather than a gain of energy. The enthalpy changes in reactions can be represented visually be graphs similar to the ones on the side. Again, the one on the left that illustrates an exothermic reaction would be most common among the reaction between food molecules and saliva that take place so that the recognition between the tastant and the receptor can occur. Additionally, it is important to note that Overall, enthalpy takes into consideration the change in energy of the system and the work done on its surroundings. Therefore; delta H = delta E + delta (PV) delta H = change in enthalpy of the system delta E = the heat absorbed by the system minus the work performed delta (PxV) = work done...however, taste will always occur at a constant pressure so it is just the change in volume that has any effect on the work When heat is added to the system during a reaction, majority of the energy is put towards doing the work to break the bonds and such (activation energy), whereas only the remaining is what goes to increasing the energy/temperature of the system. |